Neuralink is restoring speech to those who have lost the ability to speak
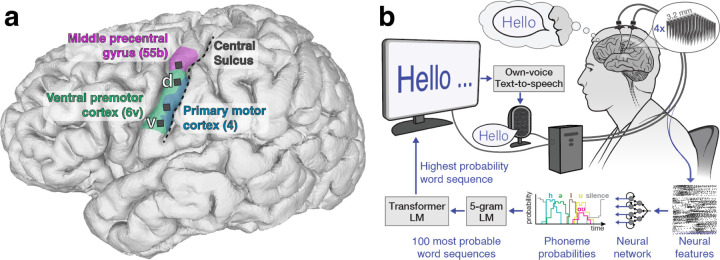
Figure 1 from a 2024 PMC preprint: left panel shows the implant/electrode locations over speech motor cortex and right panel is a clear diagram of the brain-to-text speech neuroprosthesis (neural feature extraction → phoneme decoding → language models → own-voice text-to-speech). This infographic directly illustrates the implant + decoding pipeline used to restore communication for people who cannot speak, making it highly relevant to the topic.
Source: PubMed Central (National Institutes of Health)
Research Brief
What our analysis found
Neuralink has made significant strides toward restoring communication for people who have lost the ability to speak, though the claim requires important context. In May 2025, the company announced it received FDA Breakthrough Device Designation for a speech-restoration implant, a regulatory pathway that accelerates development but does not constitute approval or proof of clinical benefit. The company's PRIME early-feasibility study, registered on ClinicalTrials.gov as NCT06429735, began enrolling participants in January 2024 with an estimated 15 participants and a primary completion target of mid-2026.
By September 2025, Neuralink reported that 12 people had received implants, accumulating roughly 2,000 days and over 15,000 hours of device use. Among them, an ALS patient named Bradford G. Smith, who had lost the ability to speak, demonstrated communication through the implant using a cursor-control-to-typing pipeline paired with text-to-speech and voice cloning — not direct neural decoding of speech. Independent academic research, including a landmark 2023 Nature study by Willett et al. achieving decoding speeds of up to ~62 words per minute, has demonstrated the broader scientific feasibility of speech neuroprostheses from cortical signals.
However, as of early 2026, no peer-reviewed, independently validated paper detailing Neuralink's human speech-restoration outcomes has been published in a scientific journal. Bloomberg reported in October 2025 that patient data had been submitted for journal review, but full methods and results have not yet undergone public scientific scrutiny. This means Neuralink's speech-restoration capabilities, while promising, remain largely unverified outside the company's own announcements and patient testimonials.
Fact Check
Evidence from both sides
Supporting Evidence
FDA Breakthrough Device Designation
In May 2025, Neuralink publicly announced it received FDA Breakthrough Device Designation specifically for a speech-restoration implant, signaling that the agency recognizes the device addresses an unmet medical need and that Neuralink is actively pursuing this indication, as reported by Reuters, Yahoo Tech, and Forbes.
Active human clinical trial
Neuralink's PRIME study (NCT
is a registered, IDE-regulated early-feasibility trial that began in January 202...
is a registered, IDE-regulated early-feasibility trial that began in January 2024, enrolling participants with tetraparesis or tetraplegia including those with ALS — a condition that frequently causes loss of speech — providing direct evidence of human testing according to ClinicalTrials.gov.
Real-world patient demonstration
ALS patient Bradford G. Smith, who had lost the ability to speak, was featured using Neuralink's implant to communicate via thought-controlled cursor typing paired with text-to-speech and voice cloning, as reported by ALS News Today, representing a concrete case of a nonverbal individual regaining communicative ability through the device.
Proven scientific feasibility from independent labs
Peer-reviewed research by Willett et al., published in Nature in August 2023, demonstrated intracortical decoding of attempted speech at up to approximately 62 words per minute, establishing that the underlying technology of restoring speech from brain signals is scientifically viable.
Significant cumulative human use data
By September 2025, Neuralink reported 12 implant recipients with approximately 2,000 cumulative days and over 15,000 hours of device use, as reported by Yahoo Finance, suggesting sustained real-world operation of the technology.
Contradicting Evidence
Breakthrough designation is not FDA approval
The FDA Breakthrough Devices Program accelerates development and review interactions but does not constitute marketing authorization or proof that the device is safe and effective for general use, according to the FDA's own program description. The designation is a procedural milestone, not a clinical endorsement.
No peer-reviewed publication of Neuralink's human results
As of early 2026, no Neuralink-authored, peer-reviewed paper reporting human speech-restoration outcomes had been publicly indexed in a scientific journal. Bloomberg reported in October 2025 that patient data was submitted for review, meaning independent scientific validation of the company's claims remains pending.
Communication method is indirect, not direct speech decoding
The demonstrated pathway involves the implant controlling a cursor to type text, which is then converted to speech via text-to-speech software and voice cloning — this is fundamentally different from directly decoding attempted speech from neural activity, making the claim of "restoring speech" potentially misleading.
Historical safety and oversight concerns
Investigative journalism, including reporting by Wired and other outlets, has documented controversial outcomes in Neuralink's animal testing phases and raised questions about the company's safety oversight practices, suggesting caution is warranted in evaluating the company's public claims.
Very small sample size and early-stage trial
The PRIME study targets only approximately 15 participants and remains in early feasibility stages with primary completion not expected until mid-2026, meaning broad conclusions about the device's ability to restore speech cannot yet be drawn from such limited data.
Report an Issue
Found something wrong with this article? Let us know and we'll look into it.